Bone remodeling associated with CTLA-4 inhibition: an unreported side effect
Background Ipilimumab is a recombinant IgG1 kappa antibody against human cytotoxic T-lymphocyte antigen 4 (CTLA-4) that augments T-cell activation. It has been approved for treating unresectable or metastatic melanoma. Preclinical investigations have recently shown that CTLA-4 inhibition can cause cytokine-mediated increase in bone remodeling.
Objective To examine whether the use of ipilumumab results in increased bone remodeling that manifests as an autoimmune reaction.
Methods A retrospective case-control study of 51 patients with stage III/IV melanoma was conducted with 2 cohorts: ipilumimab cohort at the standard dose (n = 39) and chemotherapy cohort receiving an investigational chemotherapy regimen (n = 12). Outcome variables were recorded at baseline, with each treatment, and upon treatment completion. The differences in trends of bone pain and mean alkaline phosphatase (ALP) levels were assessed using a generalized linear mixed-effect model.
Results 14 of 39 patients (35.9%) in the ipilimumab cohort reported having bone pain during at least 1 of the treatment cycles, compared with 3 of 12 patients (25%) in the chemotherapy cohort, and that trend was statistically significant over time (P = .023). Trends of mean ALP levels between the 2 cohorts were not statistically significant (P = .653). There was no correlation between bone pain or mean ALP level and response to ipilimumab.
Limitations The patient-reported bone pain was dichotomized; there was a significant variability in perceived pain intensity among patients.
Conclusions Given the increasing use of CTLA-4 inhibitors in the treatment of various malignancies, physicians should be alerted to their bone remodeling effects so that they can appropriately manage the short-term side effects such as bone pain, and monitor patients for possible unknown long-term side effects. In addition, data from postmarketing studies should be evaluated for confirmation of this novel interaction.
Accepted for publication March 22, 2017
†Drs Mansour and Rao contributed equally to the study.
Correspondence Joshua Mansour, MD; mansoour@musc.edu
Disclosures The authors report no disclosures/conflicts of interest.
Citation JCSO 2017;15(4):e217-e220
©2017 Frontline Medical Communications
doi https://doi.org/10.12788/jcso.0340
Submit a paper here
Cytotoxic T-lymphocyte antigen 4 (CTLA-4) is an important component of the immune checkpoint pathway. CTLA-4 inhibition causes T-cell activation and proliferation, increases T-cell responsiveness, and enhances the anti-tumor immune response. CTLA-4 inhibition also results in immune-related adverse reactions such as colitis, hepatitis, and endocrinopathies. Preclinical investigations have recently shown that CTLA-4 inhibition can cause cytokine-mediated increase in bone remodeling.1,2(p4) Ipilimumab, a recombinant IgG1 kappa antibody against human CTLA-4, has been approved for use in unresectable or metastatic melanoma. We hypothesize that ipilumumab results in increase in bone remodeling manifesting as an autoimmune reaction.
Methods
We conducted a retrospective case-control study of patients with stage III/IV melanoma treated at the University of New Mexico Comprehensive Cancer Center during April 2009-July 2014. The university’s Institutional Review Board approved the study.
Two cohorts were compared: an ipilumimab cohort receiving ipilumimab at 3 mg/kg every 3 weeks, and a chemotherapy cohort receiving an investigational chemotherapy regimen: carboplatin IV at an area under curve of 5 on day 1, paclitaxel IV at 175 mg/m2 on day 1, and temozolomide orally at 125 mg/m2 daily on days 2 to 6 every 21 days. Patients receiving at least 1 cycle of treatment were included. Those with known hepatic disease or concurrent malignancy were excluded from the study.
Serum ALP level (normal range, 38-150 international units per liter [IU/L]) and patient-reported bone pain measured by the 11-point numeric rating scale (NRS) for pain assessment were recorded before treatment initiation, on each cycle, and upon treatment completion.3 Clinical response was assessed per RECIST guidelines.4 Bone pain was dichotomized into Absent (pain intensity of 0 on the NRS, meaning no pain) or Present (pain intensity of 1-10 on the NRS, with 1 = mild pain and 10 = worst imaginable pain). Patients with a complete or partial response to the therapy were categorized as responders, and those with progressive or stable disease were categorized as nonresponders.
Descriptive statistics were generated for demographic and clinical characteristics. The primary outcome variables of interest were bone pain and mean ALP levels. Generalized linear mixed-effect models for proportion of patients with bone pain (with logit link function) and mean ALP levels (with identify link function) were used to evaluate for a difference in trends between the two cohorts over time. We used the Kenward-Roger approach to adjust for the small size of the degrees of freedom. To assess the significance of difference of the proportions of patients with bone pain and the mean ALP levels between responders and nonresponders in the ipilumimab cohort, the Fisher exact test and Wilcoxon rank-sum test were used, respectively. Statistical analyses were performed with statistical packages R (v3.1.3) and SAS (v9.4).
Results
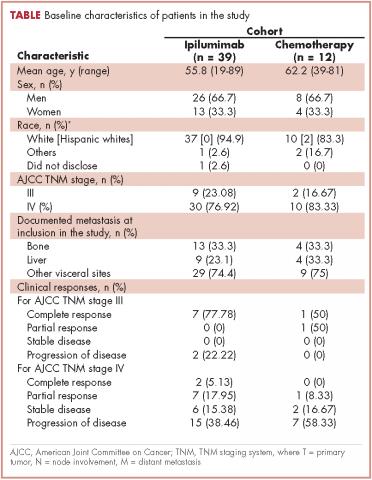
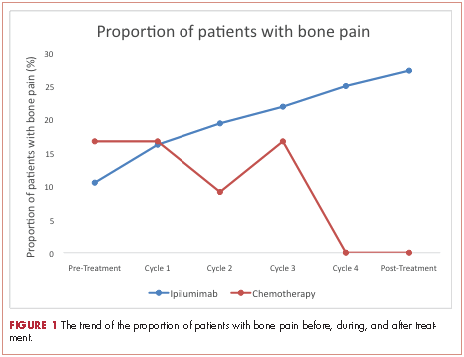
A total of 281 patients were screened, and 51 met the inclusion criteria (39 in the ipilumimab and 12 in chemotherapy cohorts). Baseline parameters were well matched between the cohorts (Table). Of the 39 patients in the ipilimumab cohort, 14 (35.9%) had bone pain during at least one of the treatment cycles, compared with 3 of the 12 patients (25%) in the chemotherapy cohort. At baseline, 4 of 38 ipilimumab patients (10.5%; 95% confidence interval [CI], 2.9-24.8) and 2 of 12 chemotherapy patients (16.7%; 95% CI, 2.1-48.4) had bone pain. Upon treatment completion, 9 of 33 ipilimumab patients (27.3%; 95% CI, 13.3-45.5) and 0 of 12 chemotherapy patients (0%; 95% CI, 0-26.5) had bone pain. The trend of proportion of patients with bone pain over time was statistically significant between the two cohorts (P = .023, Figure 1). The trends of proportion of patients with bone pain were not statistically significant when stratified by the presence of bone metastasis at inclusion in the study (P = .418) or disease progression at treatment completion (P = .500).
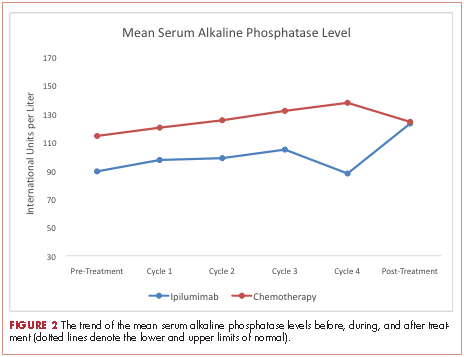
At baseline, the mean ALP level was 89.39 IU/L (95% CI, 81.03-97.75) in the ipilumimab cohort and 114.33 IU/L (95% CI, 69.48-159.19) in the chemotherapy cohort. Upon treatment completion, the mean ALP level was 123.09 IU/L (95% C.I. 80.78-165.41) in the ipilumimab cohort and 124.24 IU/L (95% C.I. 90.88-157.62) in the chemotherapy cohort. The trend of mean ALP level over time was not statistically significant between the 2 cohorts (P = .653, Figure 2).