Surgical considerations for tremor and dystonia
ABSTRACTDeep brain stimulation (DBS) is among the most effective approaches for the treatment of patients with advanced movement disorders. In patients with essential tremor, stimulation typically targets the ventral intermediate nucleus of the thalamus. Results of several studies have shown that over a follow-up period of 1 to 5 years, the severity of tremor decreases by an average of approximately 50% from baseline. Ongoing research continues to define the optimal stimulation parameters for patients with tremor, including frequency, voltage, and pulse width. In patients with dystonia, DBS typically targets the globus pallidus internus or the subthalamic nucleus. Long-term prospective clinical trials demonstrated reductions in motor severity rating scale scores of approximately 50% to 80% over follow-up periods of 2 to 3 years. Serious adverse events were uncommon, and included lead failures and infections. Appropriate candidates for DBS treatment of dystonia include patients with an unequivocal diagnosis of dystonia and significant disability. Several issues in the use of DBS for movement disorders remain unresolved, including the intensity of appropriate medical management before undergoing DBS, the importance of intraoperative mapping, optimal stimulator programming, and the time course of the beneficial effects of treatment.
Over the last decade, several studies have demonstrated that deep brain stimulation (DBS) is among the most effective approaches for the treatment of patients with advanced movement disorders, including chorea, levodopa-induced dyskinesia, tremor, and dystonia.1 The goal of DBS is to restore function or relieve pain by stimulating neuronal activity through surgically implanted electrodes. DBS produces marked and persistent reductions in abnormal movements in patients with common hyperkinetic disorders, with a generally low incidence of serious adverse events in pediatric patients and adults.
DEEP BRAIN STIMULATION FOR ESSENTIAL TREMOR
Improvement with thalamic DBS
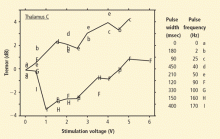
The ventral intermediate nucleus (VIM) of the thalamus is the most common target for DBS treatment of essential tremor. Several studies have demonstrated significant long-term improvement in tremor following thalamic DBS.3 Most studies enrolled 20 to 30 patients, who were followed for 1 to 5 years after device implantation. On average, these studies reported an improvement in overall tremor of approximately 50% from baseline with thalamic DBS.
Patient selection and stimulation parameters
Symptoms targeted for DBS treatment include unilateral and sometimes bilateral limb tremor. Some evidence exists for effectiveness in axial and vocal tremor as well. Factors to consider in patient selection for DBS surgery include tremor severity, degree of refractoriness to medication, and type of tremor. In addition, individual patient characteristics should be considered, including age, comorbid conditions, surgical risk, patient preference, social and employment factors, and social support.
DEEP BRAIN STIMULATION FOR DYSTONIA
Dystonia is characterized by involuntary twisting muscle contractions causing abnormal postures sometimes accompanied by jerky or repetitive involuntary movements. It may be classified according to the body part affected as generalized, segmental, or focal; in some cases it may be classified as multifocal dystonia or hemidystonia. Dystonia is also classified as primary or secondary, according to etiology. Primary dystonias are those not caused by any other identifiable condition and not associated with other neurologic abnormalities. These include idiopathic and some genetic dystonias, such as the DYT1 torsinA gene mutation. DBS of the globus pallidus internus (GPi) or subthalamic nucleus (STN) was approved by the US Food and Drug Administration under a humanitarian device exemption in 2003 for the treatment of primary generalized dystonia (PGD) in patients aged 7 years and older; GPi is the more common target).1
Evidence of efficacy
Several clinical studies have demonstrated the efficacy of DBS for patients with disabling PGD that is unresponsive to pharmacotherapy.
Long-term efficacy. Isaias and colleagues examined long-term safety and efficacy of DBS in 30 consecutive patients with PGD who were followed for at least 3 years after pallidal DBS surgery.6 DBS was delivered bilaterally in 28 patients and unilaterally in 2 patients. Clinical rating scales of motor function improved by a mean of 82.5% after 2 years, and dystonia-related disability improved by a mean of 75.2%. Improvement in motor function from baseline was noted for all 30 subjects. In five patients who were followed for 7 years, improvement in motor function remained greater than 80% at the last follow-up visit. Transient regressions were noted for patients with hardware failures or whose batteries had reached the end of life. Stimulation-related adverse events were reported for three patients and included speech difficulties and, in one patient, transient blepharospasm.
Vidailhet and colleagues examined the efficacy of bilateral pallidal stimulation in 22 patients with PGD who were followed prospectively for 3 years.7 Mean improvement from baseline in motor function on a dystonia rating scale was 51% after 1 year and 58% after 3 years (P = .03). Significant improvement was noted for individual ratings of upper and lower limb function scores. Health-related quality of life was also significantly improved at 3-year follow-up (P = .05). Serious adverse events were reported for three patients, including two lead fractures and one infection.
Results from double-blind trial. Kupsch and colleagues performed a randomized, double-blind clinical trial comparing pallidal DBS versus device implantation and sham stimulation in 40 patients with primary segmental or generalized dystonia.8 After 3 months, the mean change from baseline in severity of dystonia was 15.8% for patients who received DBS versus 1.4% with sham stimulation (P < .001). At the conclusion of the double-blind treatment phase, patients entered an open-label extension phase in which all patients received DBS for another 3 months. The initial benefit of treatment was sustained across the entire 6-month study period for patients initially randomized to DBS, whereas patients who were initially randomized to sham stimulation exhibited improved motor function during the open-label extension phase. Ratings of disability and quality of life also improved for patients receiving DBS at the end of the 6-month study. Adverse events included dysarthria (five patients), serious infections (four patients), and lead dislodgement (one patient).
Response with DYT1mutation. Coubes and colleagues examined the long-term efficacy and safety of bilateral DBS in 31 children and adults with PGD.9 PGD is associated with autosomal DYT1 mutations in approximately 30% of cases, and these authors examined the effects of treatment in patients with and without the DYT1 mutation. After 2 years of treatment, mean scores on a dystonia clinical rating scale decreased by 79% from baseline, and mean disability ratings decreased by 65%. The improvement in clinical dystonia rating scale scores was significantly greater for children than adults after 2 years (84.7% vs 70.1%; P = .04). In children, functional improvement was greater after 2 years in the subset of patients with DYT1 mutations than in the subset of patients without (76.1% vs 44.5%; P = .03), whereas in adults, DYT1 mutation status did not significantly influence response to treatment. One case of unilateral infection was noted, which required removal of the implant with successful reimplantation 6 months later. No other adverse events were reported.