Implementation of a Pharmacist-Led Penicillin Allergy Interview at a Veterans Care Facility
Background: Evaluating a patient’s Β-lactam allergy involves a thorough review of electronic health records (EHRs), penicillin skin test, and observed oral challenge. The purpose of this study was to evaluate the impact of pharmacist-led penicillin allergy delabeling solely through an interview using the Allergy to Β-Lactam Evaluation (ABLE) process.
Methods: This single-facility, retrospective cohort study reviewed EHR notes. Patient eligibility for allergy profile modifications was determined using guidance from the ABLE process. Interviews were conducted with patients meeting inclusion criteria. A progress note was entered into the EHR following completion of the interview. Patients were compared to a historical control group and matched for allergy severity (immunoglobulin E [IgE]-mediated allergy, unknown, adverse drug reaction [ADR], and severe cutaneous or other non–IgE-mediated reaction). The primary outcome was the number of patient allergy ADRs that were removed or changed on their profile.
Results: Six of 35 patients in the interviewed group had allergy profile modifications. The number of patients with a penicillin allergy/ADR modified regardless of change in their antibiotic regimen was statistically higher in the interviewed group compared with the control group: 6 (17.1%) vs 0 (0%) (P < .03).
Conclusions: The study findings support the use of the ABLE process for penicillin allergy/ADR modification or clarification. Although the sample size was small, the ABLE process has the ability to positively impact individuals with inappropriate penicillin allergy documentation in the EHR.
Self-reported penicillin allergies are common, with a prevalence of about 10% of patients, according to the Centers for Disease Control and Prevention (CDC).1 However, only about 1% of patients have a true immunoglobulin E (IgE)-mediated allergy. This issue is often further complicated by inaccurate classification of nonallergic adverse effects as an allergy, resulting in incomplete allergy documentation in the electronic health record (EHR). The cross-reactivity rate with cephalosporins (Β-lactam antibiotics) in patients reporting a penicillin allergy is < 1%, which suggests that many patients with reported penicillin allergies can safely receive them.2 Despite this, patients with self-reported penicillin allergies often receive non–Β-lactam antibiotic agents, which may be associated with an increased risk of adverse drug reactions (ADRs), increased health care costs, and inferior clinical outcomes.3
Several strategies are recommended to assess patients with self-reported penicillin allergies. According to the CDC, evaluating a patient who reports a penicillin or other Β-lactam antibiotic allergy involves 3 steps: (1) obtaining a thorough medical history, including previous exposures to penicillin or other Β-lactam antibiotic; (2) performing a skin test using the penicillin major and minor determinants; and (3) among those who have a negative penicillin skin test, performing an observed oral challenge with 250 mg amoxicillin before proceeding directly to treatment with the indicated Β-lactam therapy.4
Most existing clinical guidance for assessing patients with self-reported penicillin allergies stems from site-specific policies and primarily focuses on oral amoxicillin challenges or penicillin skin testing (PST). However, performing these tests may not be feasible at all facilities due to time constraints and lack of allergists. Therefore, alternative strategies are necessary, such as conducting detailed patient interviews. Few studies have evaluated switching to Β-lactam agents following a penicillin allergy interview alone. However, with thorough patient histories and detailed interviews, patients with reported penicillin allergies can safely use Β-lactam antibiotics.5 Implementing this procedure provides a cost-savings opportunity by not having to administer additional antibiotics for testing in addition to improving antibiotic stewardship.
The Memphis Veterans Affairs Medical Center (MVAMC) created the Allergy to Β-Lactam Evaluation (ABLE) process to clarify and remove penicillin allergies. The process involves conducting a thorough chart review and patient interview followed by completion of a note template that provides recommendations about patient allergies and Β-lactam prescribing. Mitchell et al found that the pharmacist-led process to be beneficial for addressing Β-lactam allergy clearance.6 As a result, the ABLE process was implemented at several other US Department of Veterans Affairs (VA) medical centers (VAMCs). Using the ABLE template, the purpose of this study was to evaluate the impact of a pharmacist-led penicillin allergy initiative on penicillin allergy delabeling with an interview process alone.
Methods
Prior to ABLE process implementation, there were no standardized procedures for documenting allergy histories. ABLE was implemented at the Robley Rex VAMC (RRVAMC) in November 2022. During the interview phase, patients were initially identified during admission via TheraDoc as having either a penicillin allergy or ADR. The infectious disease pharmacist or pharmacy resident interviewed patients with documented penicillin allergies or ADRs using a standardized questionnaire (eAppendix 1). Not all identified patients could be interviewed. Patients currently receiving an antibiotic were prioritized for interviews. Patients were excluded if they declined or were unable to be interviewed, although a patient’s caregiver(s) could be interviewed in person or via telephone, if the patient was not available.
Following the interview, pharmacists used guidance from the ABLE process in addition to a detailed EHR review to determine whether the patient was eligible for an allergy update or removal and/or switch to a Β-lactam antibiotic (Figure). If eligible for modification, the interviewing pharmacist made the necessary changes. A templated process note with patient-specific recommendations was entered into the Computerized Patient Record System (CPRS) and the primary care team attending physician was added as an additional signer to be alerted in the system note (eAppendix 2).
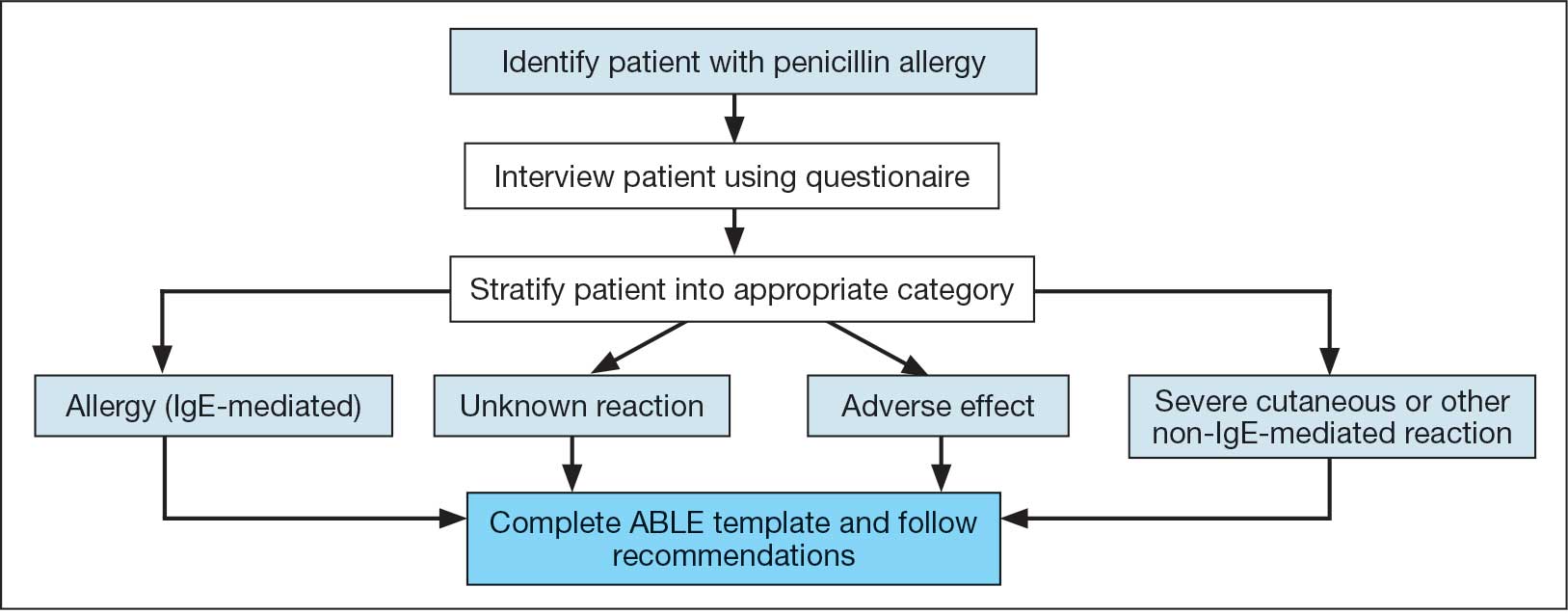
This single-center, retrospective cohort study involved review of CPRS notes and clinical interviews in the interviewed group. Hospitalized patients at the RRVAMC aged ≥ 18 years with a documented penicillin allergy or ADR were included. The historical control group consisted of patients admitted between October 31, 2019, and October 31, 2022, and the intervention group consisted of patients admitted between November 1, 2022, and March 1, 2023. Patients in the historical control group were matched 1:1 to the intervention group for penicillin allergy severity (allergy [IgE-mediated], unknown, adverse effect, severe cutaneous or other non–IgE-mediated reaction) and whether they received a noncarbapenem non–Β-lactam antibiotic.
The primary outcome was the number of patient allergies/ADRs removed or changed on patient profiles regardless of whether their antibiotic regimen was changed. This outcome was further assessed by evaluating the number of patient allergies or ADRs removed or changed on patient profiles with or without a change in antibiotic regimen. Primary outcomes were analyzed using χ2 and/ or Fisher exact tests, as appropriate to determine statistically significant differences between the interviewed group and the historical control.
Results
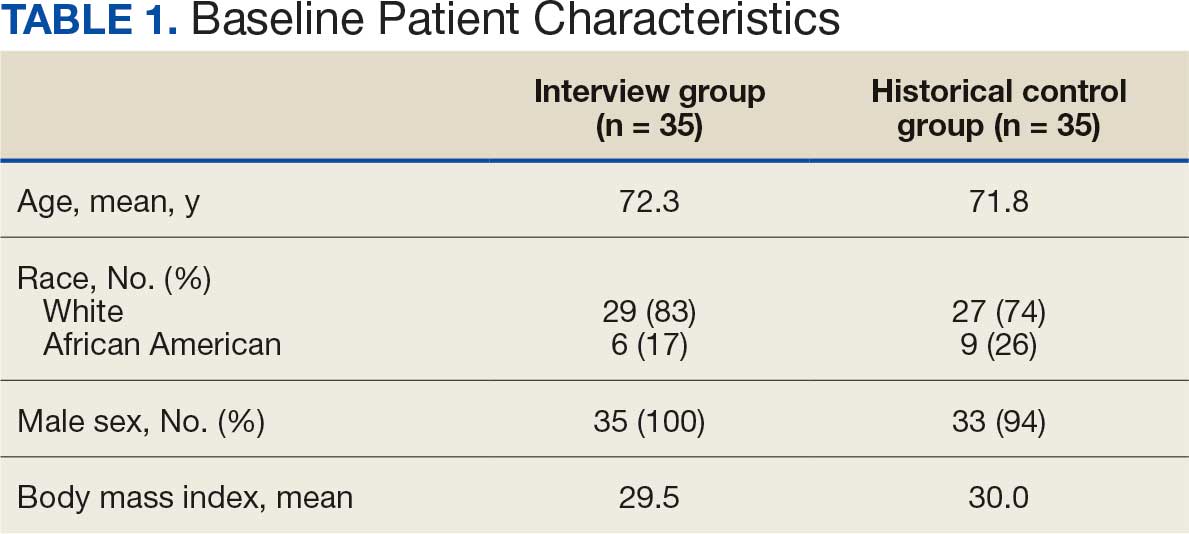
Seventy patients were included: 35 patients in the interviewed group and 35 patients in the historical control group, respectively. Both groups had a mean age of 72 years and predominantly included White male patients (Table 1). Following the interview, the allergy profile was modified for 6 patients (17%) in the interview group vs 0 patients in the control group (P = .03) (Table 2). The primary outcome was analyzed separately regardless of an antibiotic regimen change. There was not a statistically significant difference between groups when assessing patients for change in therapy (P > .99). All 6 patients with an allergy profile modification had no change in antibiotic regimen.

Discussion
This study suggests the ABLE process may be a valuable tool for adjusting penicillin allergies or ADRs within patient EHRs. In the interview group, allergies were modified in 6 (17%) patients while no patients in the control group had allergy modifications. Of the 6 allergy profile modifications, 4 allergy labels were changed from an allergy to an ADR. These patients were cleared to receive future Β-lactam antibiotics after clinicians recognized the lack of a true IgE-mediated allergic reaction. In addition, 2 of the modified allergy profiles removed the allergy designation. Although this represents a small subset of interviewed patients, it illustrates the clinical effectiveness of an interview process alone to remove penicillin allergy designations.
Previous research has assessed the impact of pharmacist intervention on penicillin allergy clarification. Mitchell et al implemented a pharmacist-driven Β-lactam allergy assessment and penicillin allergy clinic (PAC) at the MVAMC with the goal of evaluating its impact on allergy clearance. In their study, clinical pharmacy specialists evaluated patients with Β-lactam allergies, and those deemed eligible were later seen in the PAC. Among the 246 patients evaluated using the Β-lactam allergy assessment alone and who were not seen in the PAC, 25% had their penicillin allergy removed following a detailed assessment.6
Song et al evaluated the effectiveness and feasibility of a pharmacist-driven penicillin allergy delabeling pilot program without skin testing or oral challenges. Patients with penicillin allergies were interviewed by a pharmacy resident using a standardized checklist. Among the 66 patients interviewed, 12 (18%) met the criteria for delabeling and consented to removal of their allergy.7 The delabeling rates in these 2 studies are similar to the 17% rate of allergy modification in our study, although this study is the only one to compare results to a historical control group.
Harper et al evaluated the impact of a penicillin allergy assessment, including penicillin skin testing and oral amoxicillin challenges, on delabeling penicillin allergies. Pharmacists completed a penicillin allergy assessment and performed penicillin skin testing and/or oral amoxicillin challenges for eligible patients. Of 35 patients, 31 (89%) had their penicillin allergies delabeled in the EHR.8 The rate of penicillin allergy delabeling in Harper et al was likely higher than that seen in our study due to the use of oral challenge and skin testing. Regardless, a detailed penicillin allergy interview alone was effective at RRVAMC, resulting in a significant rate of allergy removal or change. This supports the use of detailed penicillin allergy assessments in settings where penicillin skin testing or oral challenges may not be feasible.
Mann et al demonstrated the effectiveness of penicillin allergy assessments in switching eligible patients to Β-lactam antibiotics. Their single-center, prospective study assessed the impact of a pharmacist-driven detailed penicillin allergy interview initiative. Interviews that evaluated potential changes to allergy profiles were conducted with 175 patients. Of these patients, 135 (77.1%) were on antimicrobial therapy and 42 (31.1%) patients receiving therapy met criteria to switch to a noncarbapenem Β-lactam antibiotic. Thirty-one patients (73.8%) switched with no signs or symptoms of intolerance demonstrating that an interview can be a valuable tool for antibiotic optimization, specifically in patients with penicillin allergy.9 No patients in our study switched antibiotic therapy, likely because only a small number of patients were eligible for transition to a noncarbapenem Β-lactam antibiotic. In the Mann et al study, non–Β-lactam antibiotics, such as fluoroquinolones and carbapenems, accounted for > 75% of the antibiotics used.
Limitations
The sample size of this study was small and its duration was short. There is a risk for selection bias as not all identified patients were able to be interviewed while admitted, but patients on antibiotics were prioritized as they were most likely to directly benefit during their current admission from a modification of their allergy. Most patients in the study were White and male, which may limit the generalizability of the results. Additionally, recommendations regarding antibiotic changes were primarily communicated to the treatment team based on a templated note in CPRS alone. Therefore, implementation of these recommendations largely relied upon nonverbal communication. Direct pharmacist-physician communication could have led to a larger impact on antimicrobial therapy changes. The interviewer’s participation in daily rounds with time allotted to discuss this topic can be considered in the future to improve these processes.
Conclusions
This study found that the ABLE process identified patients for penicillin allergy delabeling. With the high prevalence of inaccurate penicillin allergy documentation, this tool offers VA health care systems a way to empower pharmacists in allergy clarification, leading to improvements in antibiotic stewardship. Although the sample size was small, the ABLE process may provide a framework for VA clinicians. Future research has the potential to demonstrate the practicality and effectiveness this pharmacist-led penicillin allergy interview process can offer clinicians.




